Table of contents
1. The Challenge: Cervical Cancer and Traditional Screening Methods
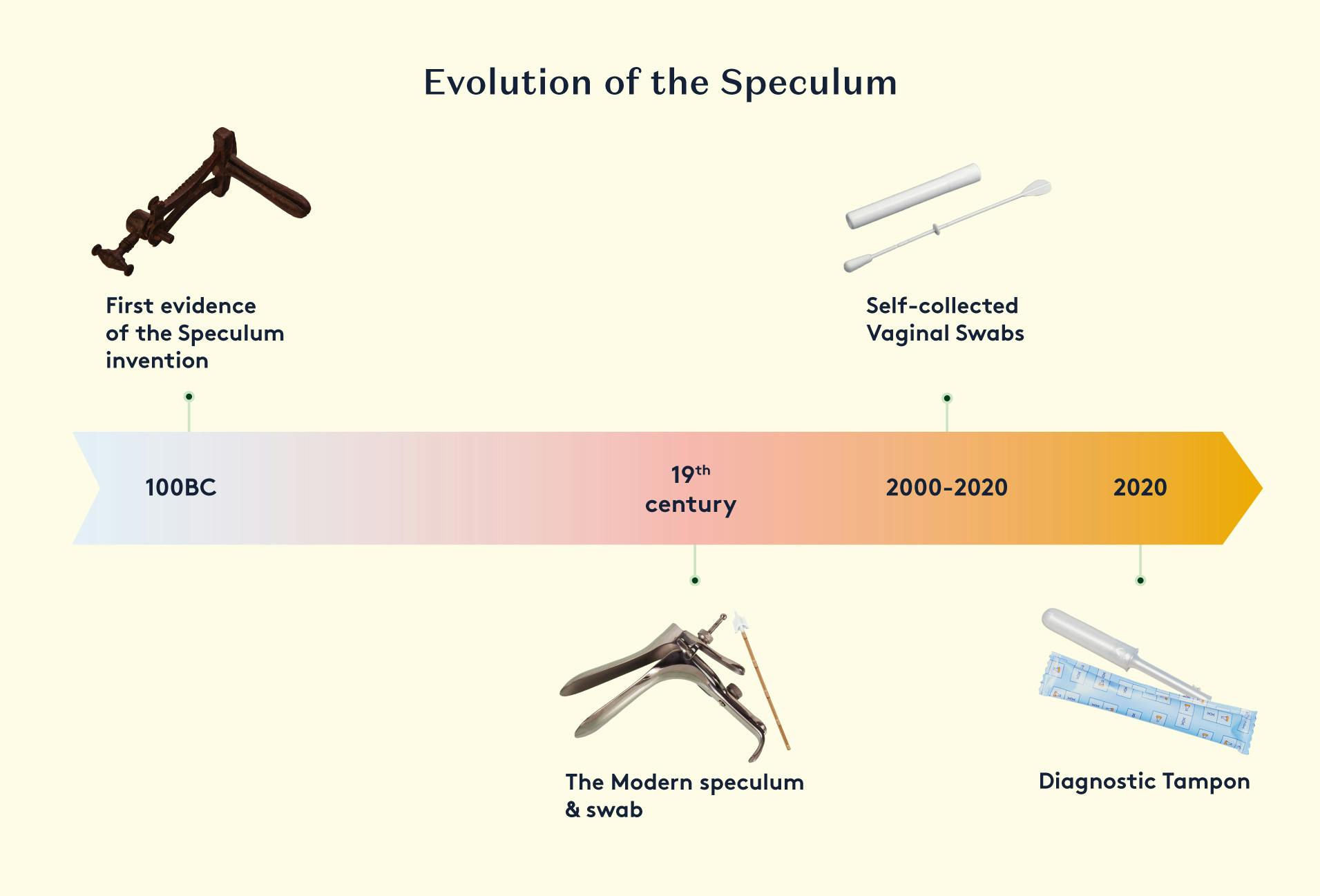
2. Daye's Solution: The Diagnostic Tampon
3. Recognition by the NHS
4. A Shot at Eradicating Cervical Cancer
5. Together, We Can End Cervical Cancer by 2040
Written by Valentina Milanova
Illustrated by Maria Papazova
At Daye, we are thrilled to announce that we have been invited to join the prestigious NHS Innovation Accelerator program for a three-year term. This partnership is a significant milestone in our mission to close the gender care gap, particularly in the fight against cervical cancer. Our innovative Diagnostic Tampon for HPV screening has been recognized as a preferred cervical cancer prevention device by the NHS, marking a crucial step towards supporting the ambitious public health goal of eradicating cervical cancer by 2040.
The Challenge: Cervical Cancer and Traditional Screening Methods
Cervical cancer remains a major global health issue, with over 570,000 new cases annually. It is primarily caused by certain types of human papillomavirus (HPV), making HPV testing essential for early detection and prevention. Despite being 99.8% preventable, cervical cancer still claims approximately 1,000 lives annually in the UK alone, with 3,900 new cases diagnosed each year.
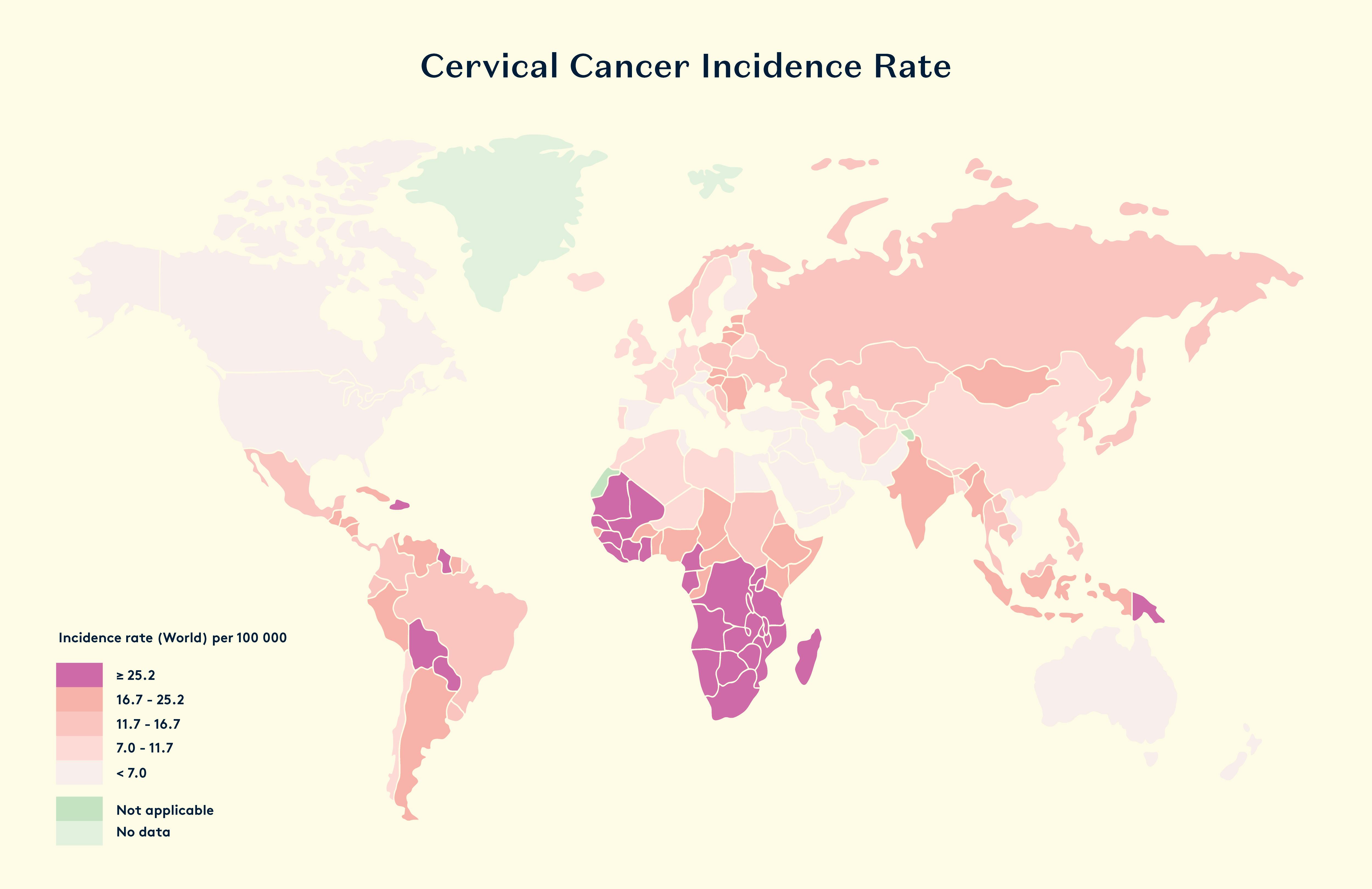
Traditional screening methods can be time-consuming, costly, invasive, and uncomfortable, leading to low participation rates and a rise in cervical cancer cases. Currently, only 62.7% of eligible individuals in the UK are adequately screened, well below the 80% target. As a result, cervical cancer mortality is predicted to rise 17% by 2030. The current NHS screening pathway, involving the pap smear, is often seen as a hassle and easy to put off due to discomfort and the invasive nature of the procedure.
In the context of an already over-stretched NHS facing 43,339 nursing staff shortages (10.9% of all nursing posts), the potential for cost savings through reduced clinic visits and staff time makes self-sampling methods like Daye's Diagnostic Tampon particularly valuable.
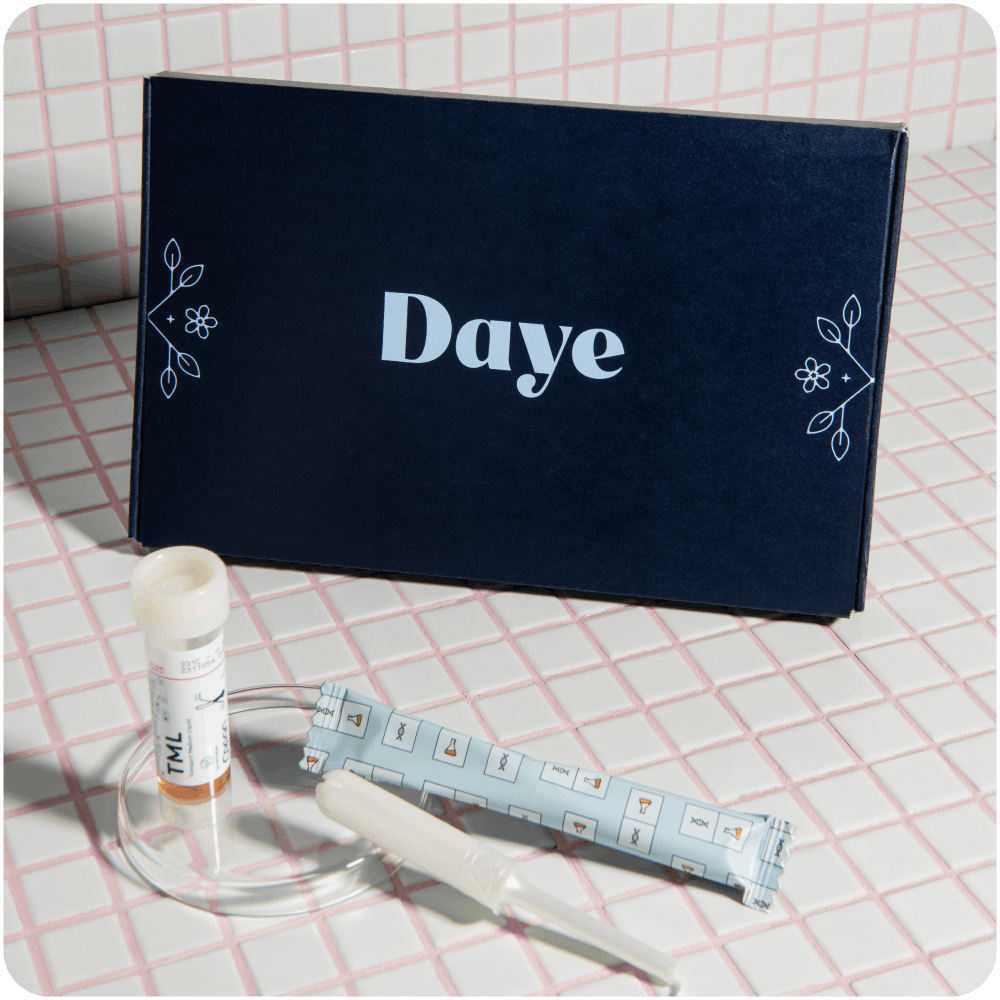
Daye's Solution: The Diagnostic Tampon
Our Diagnostic Tampon offers an affordable, patient-friendly alternative for HPV screening in the context of cervical cancer prevention. By leveraging the familiarity of tampons, our device increases comfort during self-sampling, decreases screening costs, and incentivizes more patients to participate in cervical cancer prevention programs.
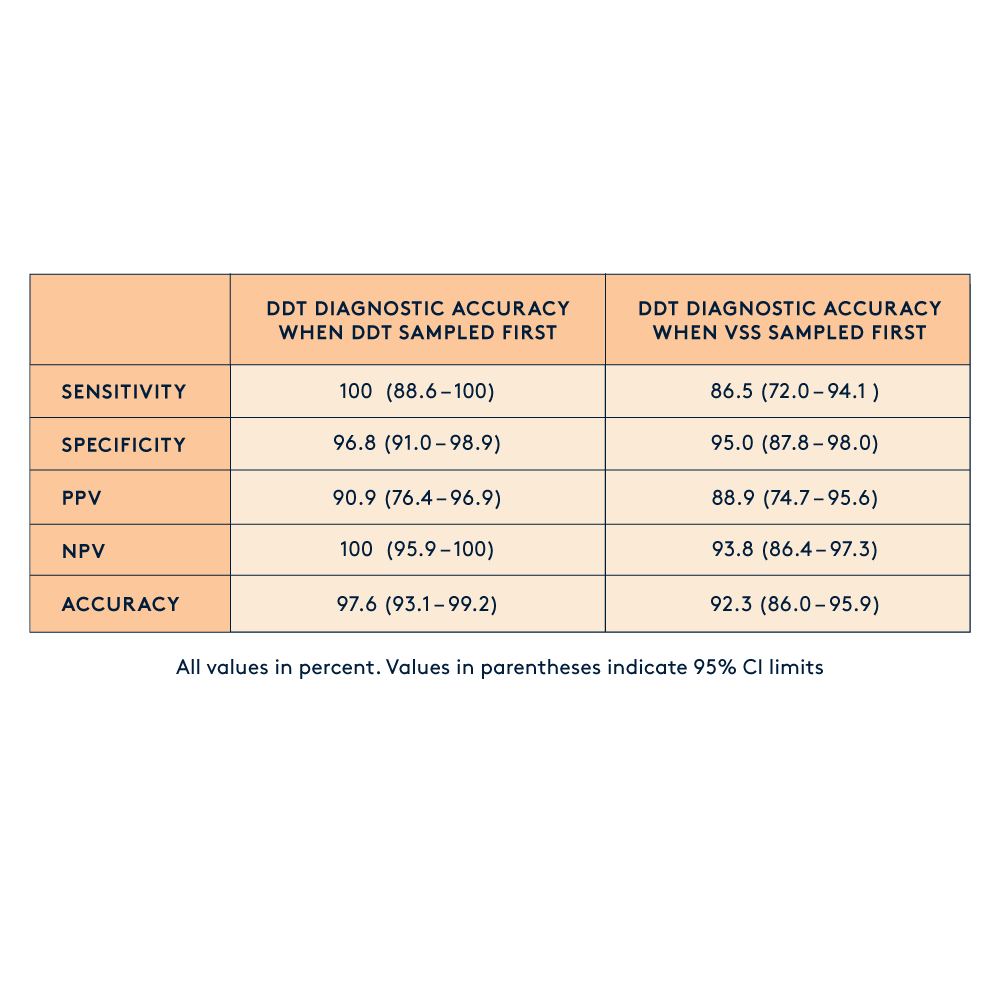
Clinical trials with statistically significant patient populations have shown that the Diagnostic Tampon achieves diagnostic accuracy comparable to clinician-collected swabs. Notably, our Diagnostic Tampon outperforms the self-collected swab.
Our innovative approach has the potential to expand screening access, particularly in low-resource settings or among individuals who avoid traditional pelvic exams, such as sexual assault survivors, transgender patients, and ethnically diverse patient groups. Daye's clinical investigations, conducted with The University of Edinburgh, NHS Lothian, and the Scottish HPV Reference Laboratory, demonstrated a 70.5% preference for the Diagnostic Tampon over the clinician-collected and the self-collected swab in focus groups. This preference is crucial for increasing screening participation rates, especially among underserved populations.
Diagnostic accuracy Diagnostic Tampon (DDT) and Vaginal Self Swab (VSS)
Recognition by the NHS
The NHS's selection of Daye's Diagnostic Tampon as a preferred device for cervical cancer screening underscores our potential to transform the public health landscape. This recognition is a testament to our commitment to developing accessible, reliable, and comfortable health solutions, which fit conveniently into patients' lives.
By integrating our technology into the NHS system, we aim to increase screening participation rates, address disparities in healthcare access, and ultimately contribute to the eradication of cervical cancer by 2040 - a goal which can only be achieved with increased access to self-sampling for HPV and a higher uptake of the HPV vaccine.
A Shot at Eradicating Cervical Cancer
We stand at the threshold of a remarkable opportunity: eradicating cervical cancer within our lifetime. The World Health Organization has set an ambitious goal to eliminate cervical cancer by the end of the century, and with innovations like the Diagnostic Tampon, we are closer than ever to achieving this vision.
By increasing screening participation and addressing disparities in healthcare access, we can significantly reduce the incidence of cervical cancer. The NHS's goal of eradicating cervical cancer by 2040 is ambitious, but with the right tools and partnerships, it is achievable.
Our technology has the potential to bridge the screening gap by making testing more accessible and appealing to those who have been underserved by traditional methods, while saving costs for the NHS. With a user-friendly device like the Diagnostic Tampon, we can increase screening rates, detect more cases of high-risk HPV, and ultimately save lives.
Together, We Can End Cervical Cancer by 2040
By joining the NHS Innovation Accelerator program, Daye will work closely with the NHS to integrate our Diagnostic Tampon into the healthcare system. This partnership aims to enhance the accessibility and acceptability of cervical cancer screening, addressing the barriers that hinder participation in traditional methods. We are committed to ensuring that our technology is not only effective but also accessible and sustainable, aligning with the NHS's vision for a healthier future.
Daye's invitation to the NHS Innovation Accelerator marks a significant step forward in the fight against cervical cancer. By combining innovative technology with user-centric design, we are poised to make a lasting impact on gynaecological health. As the NHS continues to strive for cervical cancer elimination, collaborations like this will be crucial in achieving this ambitious goal. At Daye, we are dedicated to empowering patients with accessible, reliable, and comfortable health solutions, and we look forward to this exciting journey ahead.






